What Is Chemical Makeup Of 40 Rock
When producers pay special attention to managing phosphorus (P), it tin lead to profitable crop production. The best mode to employ fertilizers to meet P requirements changes with crop, soil properties and environmental atmospheric condition.
Finding the best P source
Inorganic commercial P fertilizers accept evolved over the last several decades into a refined, predictable product. Plus, at that place are the organic P sources closely associated with livestock operations or with proximity to major metropolitan areas.
In that location should exist no divergence in P fertilizer sources, as long as nutrient assay differences are taken into account. While there are certain situations where one product performs better, phosphorus fertilizer recommendations are the same regardless of the phosphate fertilizer source.
How commercial phosphate fertilizer is manufactured
|
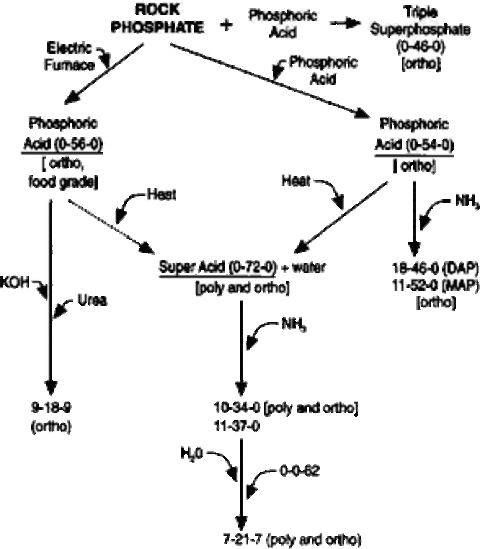
Stone phosphate is the raw textile used to manufacture most commercial phosphate fertilizers on the market place.
In the past, footing rock phosphate itself has been used as a source of P for acrid soils. However, very little stone phosphate is currently used in agriculture due to low availability of P in this native material, high transportation costs and modest ingather responses.
Nearly commercial phosphate fertilizer manufacturing begins by producing phosphoric acid.
The generalized diagram in Figure ane shows the steps taken to manufacture diverse phosphate fertilizers. Phosphoric acid is produced past either a dry or wet process.
Dry out vs. moisture process
In the dry process, an electrical furnace treats stone phosphate. This treatment produces a very pure and more expensive phosphoric acid – oft called white or furnace acid – primarily used in the nutrient and chemical industry.
Fertilizers that employ white phosphoric acid equally the P source are more often than not more expensive considering of the costly treatment process.
The moisture process involves treating the rock phosphate with acid-producing phosphoric acid – too called dark-green or black acid – and gypsum, which is removed equally a by-product. The impurities that give the acid its colour haven't been a problem in the production of dry fertilizers.
Orthophosphoric acrid
Both the moisture and dry treatment processes produce orthophosphoric acid, the phosphate form that'due south taken up by plants.
The phosphoric acrid produced by either the wet or dry process is ofttimes heated, driving off water and producing a superphosphoric acid. The phosphate concentration in superphosphoric acid normally varies from 72 to 76 per centum.
The P in this acid is nowadays every bit both orthophosphate and polyphosphate. Polyphosphates consist of a series of orthophosphates that have been chemically joined together. Upon contact with soils, polyphosphates revert back to orthophosphates.
Adding ammonia
Ammonia can exist added to the superphosphoric acrid to create liquid or dry materials containing both nitrogen (Due north) and P. The liquid, 10-34-0, is the near common product.
The 10-34-0 can exist mixed with finely basis potash (0-0-62), h2o and urea-ammonium nitrate solution (28-0-0) to form 7-21-seven and related grades. The P in these products is nowadays in both the orthophosphate and polyphosphate form.
When ammonia is added to phosphoric acid that hasn't been heated, it produces monoammonium phosphate (eleven-52-0) or diammonium phosphate (18-46-0), depending on the ratio of the mixture. The P nowadays in these two fertilizers is in the orthophosphate form.
Toll and effect
The cost of converting stone phosphate to the individual phosphate fertilizers varies with the process. More importantly, the processes have no consequence on the availability of P to plants.
Phosphate fertilizer terminology and sources
Selecting a phosphate fertilizer can be confusing due to all the products on the market. Understanding the terminology may help avert some of the defoliation.
|
Fertilizer samples analyzed past a control laboratory are placed in water, then the percentage of the total phosphate that dissolves is measured. This per centum is referred to as h2o-soluble phosphate.
The fertilizer material that isn't dissolved in water is then placed in an ammonium citrate solution. The corporeality of P dissolved in this solution is measured and expressed equally a percentage of the total in the fertilizer material.
Phosphate measured with this analytical process is referred to as citrate-soluble.
The sum of the h2o-soluble and citrate-soluble phosphates is considered to be the per centum that's available to plants and is the amount guaranteed on the fertilizer characterization. Commonly, the citrate- soluble component is less than the water-soluble component.
Comparison chart: Mutual fertilizer sources
Tabular array 1: Percentages of water-soluble and available phosphate in several common fertilizer source
Organic phosphorus sources
Organic P fertilizers take been used for centuries every bit the P source for crops. Even with the advent of P fertilizer technology processes, organic P sources from animal manures – including composts – and sewage sludge are nonetheless very of import.
|
From a fertilizer/food management perspective, the major differentiating gene is the availability of P.
Equally with any fertilizer products, specially those with varying analysis, do a chemical assay. Then apply an availability coefficient to determine the available P as a portion of the reported total P.
Phosphorus from manure or sludge should be comparable to P from inorganic fertilizer. And so, if a producer has a P recommendation for thirty pounds per acre of P<sub>2</sub>O<sub>5</sub>, applying approximately 65 pounds of 18-46-0 (DAP) or vi tons of 11-6-9 (manure; 80 percent bachelor P coefficient) should provide equivalent results.
The P contained in organic P sources combines inorganic and organic P. Essentially, all inorganic P is in the orthophosphate course, which is the class taken upwardly by growing plants.
Chemical makeup
Diet fed to the brute has some control over this chemical makeup. Consider P feed supplements and the fact that many could be considered P fertilizers also. By and large, 45 to 70 percent of manure-P is inorganic P. Organic P constitutes the remaining total P.
Decomposition
Much of the organic P is hands decaying in the soil, only factors such as temperature, soil moisture and soil pH all have a bearing on the P mineralization rate. The terminal decomposition production is orthophosphate P compounds.
Available P
The combination of the organic-inorganic P ratios in the organic P sources and the soil environment impact the availability coefficient for organic P. Most creature manure research interpretations indicate that approximately 60 to 80 percentage of the full P is available to crops in the start year.
Due to the chemical limerick of other organic P sources such as bone repast, expect lesser amounts of constitute-available P compared to total P.
How crops respond to phosphate fertilizers
If the level of bachelor P in the soil isn't adequate for optimum crop growth, use phosphate fertilizers to ensure adequate amounts of this nutrient in the solution phase.
|
Optimal soil exam levels
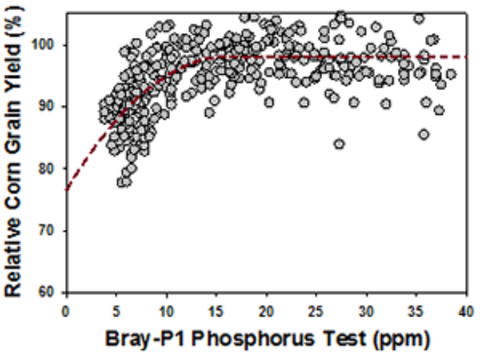
Numerous inquiry projects have demonstrated that agronomic crops volition answer to phosphate fertilization if soil test levels are in the very depression, low and medium ranges, or below 15 parts per million (ppm) in the Bray-1 test (Figure 2) or 11 ppm in the Olsen exam.
Crop removal
Crop removal is common in many areas of the state. In the instance in Table 2, banding the P at a lower rate resulted in the same yield every bit those based on crop removal recommendations.This illustrates the effect that banding P can have on reducing the corn crop's overall P requirements.
Application method
Table two: How banded starter and broadcast phosphate bear on corn yield
Shows the effect of banded starter (two inches beside and beneath the seed) and broadcast phosphate on corn yield when soil test levels for phosphorus are medium. Data are an boilerplate of two years of data nerveless at the West Key Enquiry and Outreach Center in Morris.
The ability of the banded fertilizer awarding to supply a ingather's entire P requirement can depend on the type of band used and the soil examination. Banding liquid fertilizer on the seed is mutual for corn and sugarbeet.
When banding on the seed, utilize a low rate. This is because there's potential to reduce emergence due to high salts or ammonia forming almost the seed.
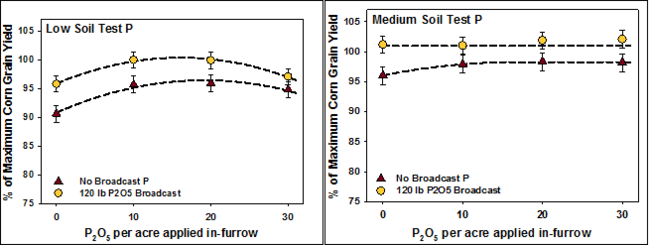
The example in Figure 3 shows that a small rate of phosphate banded with the seed can provide maximum yield for corn with medium soil test P levels. Withal, it'south not enough to maximize yield with low soil test P levels.
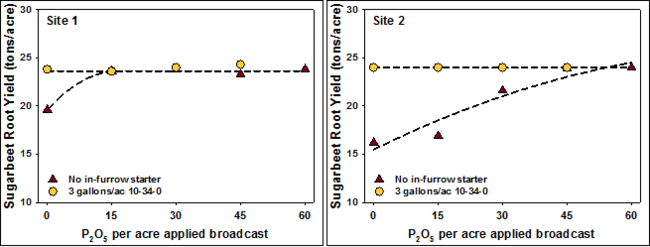
In contrast, contempo information has shown that a small rate of fertilizer banded with the seed is better than higher rates of broadcast P for sugarbeet (Figure 4).
Table ii: How banded starter and broadcast phosphate affect corn yield
The ability of the banded fertilizer awarding to supply a crop's entire P requirement can depend on the type of band used and the soil test. Banding liquid fertilizer on the seed is mutual for corn and sugarbeet.
When banding on the seed, use a low charge per unit. This is because there'southward potential to reduce emergence due to high salts or ammonia forming near the seed.
The example in Figure 3 shows that a pocket-size rate of phosphate banded with the seed can provide maximum yield for corn with medium soil exam P levels. Still, it's not enough to maximize yield with depression soil test P levels.
In dissimilarity, contempo information has shown that a small rate of fertilizer banded with the seed is amend than higher rates of broadcast P for sugarbeet (Figure iv).
Table 3 shows corn and soybean plants' response to using phosphate.
Soil test values
This case illustrates the event that starting soil examination level, soil type and crop tin have on the response to P.
Corn grain yield responded to P at two of the locations, Lamberton and Morris, while soybean only responded at Morris, which had the lowest starting soil test value for P. Agreement which crops respond better at which soil test values is of import to ensure maximum return on investment when applying P.
Application method
Tabular array 3: How the rate of phosphate circulate applied affects corn and soybean yield
Crop response to P application varies:
-
Alfalfa: Will respond to levels upwards to 40 ppm (soil test Bray P-one value).
-
Wheat and soybean: Will only respond upwardly to x to 15 ppm (soil test Bray P-i value).
-
Corn: Volition respond to levels up to 15 to xx ppm.
-
White potato: Volition respond to levels in a higher place 30 ppm. Nonetheless, response is more probable when soil test P is below 30 ppm.
Predicting the demand for phosphate fertilizer
Phosphorus soil tests measure soil's power to supply P to the soil solution for plant use, but do not measure out the total quantity of available P. These tests provide an availability index of P in soils that relates to the phosphate fertilizer's ability to provide an economically optimal increase in yield.
The human relationship between the P determined past a soil test and the phosphate fertilizer requirements are developed from the results of numerous enquiry trials that measured various rates of practical phosphate and yields.
|
Table 4 summarizes recent data on corn response to P in Minnesota. For various starting soil test values, Table 4 gives:
-
The percentage of times that applying P resulted in a measurable increase in corn yield.
-
The average yield achieved when no P was practical.
Information technology's of import to note that there's ever a possibility that applying P volition increment the crop's yield. Equally shown in Table 4, applying P in the high and very loftier categories increased corn grain yield 14 and nine percent of the fourth dimension, respectively.
However, the boilerplate yield produced in those categories was within 1 pct of the maximum of maximum. Maintaining high to very high soil examination levels will ensure maximum yield potential, but the low probability of response to P volition result in a poor economic return from high rates of practical P.
Table 4: Corn grain yield response to practical P fertilizer based on soil examination category
Olsen and Bray-1 procedures
Ii laboratory procedures are used to measure the P status of Minnesota soils:
-
Olsen process: Use when the soil pH is 7.iv or greater.
-
Bray-i procedure: Use when the soil pH is less than 7.four.
Both soil tests have been correlated and calibrated with yield response. The phosphate recommendations in Minnesota are based on those correlation values.
Some soil testing laboratories clarify soils with both a weak Bray (P-1) and a strong Bray (P-2) process. Bray P-2 results have non been correlated and calibrated to the ingather response to phosphate fertilizer in Minnesota and aren't useful in predicting the amount of phosphate fertilizer to apply.
There are several situations where the soil pH is greater than 7.four and the P value from the Bray-ane procedure is greater than the P value from the Olsen procedure.
When soil samples are analyzed past both the Olsen and Bray-1 procedures, enquiry information indicates that phosphate fertilizer recommendations should be based on the greater value. Yous can also utilise plant analysis as an aid in determining the availability of P in soils.
Mehlich-three soil test
Several states in the Corn Belt use the Mehlich-iii soil test, but it's non recommended in Minnesota.
The Mehlich-3 soil test will typically event in soil P test levels 0 to v percent greater than the Bray-P1 test when soil pH is 7.5 or less. The Mehlich-3 test has been found to be less reliable for soils with excess carbonates and a pH greater than 7.5.
Symptoms of P deficiency aren't obvious or easily identifiable for most crops in Minnesota. For nigh crops, a shortage of P reduces plant size.
Less establish growth
Figure 5 shows less constitute growth due to a shortage of P in potatoes. This lack of growth is typical for crops such as potato and soybean when P is scarce.
Purpling
For corn, a severe P deficiency inhibits the translocation of carbohydrates inside the plant. This leads to a purple colour on the margins of the leaves.
The purpling is normally most evident in young corn plants because there's a greater demand for P early in the growing season. Figure half dozen shows a P-deficient corn found.
Some hybrids have a purple advent early on in the growing season regardless of the P supply in the soil. This tin be a genetic response to stress caused by cold temperatures. Don't misfile this hybrid characteristic with P deficiency.
When to use institute analysis every bit a direction tool
It'south of import to relate the interpretation of the belittling results to the growth phase. The concentration of P in plant tissue unremarkably decreases as the found matures. Table five summarizes some interpretations of P concentrations for several crops.
Table 5: Sufficiency levels of phosphorus for major agronomic crops, vegetables and fruits grown in Minnesota
Managing phosphate fertilizers
Because P isn't mobile in soils, placing phosphate fertilizers is a major management decision in crop production systems. There's no special placement that's platonic for all crops. Decisions near placing phosphate fertilizers are primarily afflicted past the intended ingather and P soil test level.
|
For corn and small grain production, the needed phosphate fertilizer tin be:
-
Broadcast and incorporated before planting.
-
Applied in a band away from the seed row as a starter fertilizer at planting.
-
Directly on the seed at planting, if small amounts are needed.
With pocket-size grains, you can utilise the amount of needed phosphate with a drill or air seeder at planting. Corn starter fertilizer is unremarkably separated from the seed by approximately i inch of soil.
The banded application is a very efficient way to apply phosphate fertilizer, as yous can cut the recommended broadcast application rates in half.
Results advise you lot can place a pocket-sized amount of fertilizer directly on the corn seed with the planter. However, the rate applied may not satisfy the amount needed for corn if soil test phosphorus is depression.
Soybean research trials have shown that greater grain yields are produced if the needed phosphate is broadcast and incorporated before planting, compared to a ring application.
This response is the opposite of corn and minor grain, and may best exist explained by differences in the evolution of the respective root systems.
For sugar beet, electric current research suggests that seed row placement of 15 pounds of phosphate will produce similar yields every bit 45 to 60 pounds of phosphate broadcast to the soil.
For other row crops, there isn't plenty research to propose a preferred method of phosphate placement.
Applying phosphate for alfalfa and other provender crops is more efficient when done earlier stand up institution, when the fertilizer can be incorporated prior to seeding.
Grasses and legumes develop a large number of small roots most the soil surface. This ways these crops tin can blot phosphate fertilizers that are annually broadcast to established stands, if additional fertilizer is required.
Oft asked questions: Phosphate fertilizers
|
The fashion plants use P isn't affected by the liquid or dry property of the fertilizer. Establish food utilise in both liquid and dry fertilizers is affected by factors such equally:
-
Method of application.
-
Crop and root growth characteristics.
-
Soil examination levels.
-
Climatic conditions.
The corporeality of water in a fluid fertilizer is insignificant compared to the water already present in the soils. Therefore, P in liquid P sources is not more than available than P in dry materials — even in a dry year.
Base of operations your selection of a liquid or dry P source on adaptation to your farm's functioning and economics.
To answer this question, it'south of import to sympathize the departure between these two forms of phosphorus. The phosphorus in the phosphoric acid used to make most dry phosphate fertilizers as well as a few liquids is in the orthophosphate form.
Process: Manufacturing and soil conversion
If ordinary phosphoric acrid is heated, water is removed and the orthophosphate ions combine to class a polyphosphate.
This process does not catechumen 100 percent of the orthophosphate ions into the polyphosphate form. Most polyphosphate fertilizers volition take 40 to 60 percent of the phosphorus remaining in the orthophosphate grade.
In the soil, polyphosphate ions readily convert to orthophosphate ions in the presence of soil water. This conversion is rapid and, with normal soil temperatures, can be completed in days or less. An enzyme called pyrophosphatase, which is arable in about soils, enhances this conversion process.
Comparing products
Polyphosphates are usually marketed as liquid ammonium polyphosphate fertilizers. Because water is removed in the manufacturing process, these materials have a higher analysis than materials with phosphate in the orthophosphate form.
Polyphosphate liquids are also more convenient for the fertilizer dealer to handle and let for the conception of blends that aren't possible with the orthophosphate liquids.
Outcome on yield
Numerous field trials have evaluated how orthophosphate and polyphosphate fertilizers touch crop production. The results shown in Table 6 are typical of the results obtained from several trials.
Table half-dozen: How P source influences corn yield
The yields shown in Table six are averages from five sites where the soil pH was more than 7.three.
It's obvious that the form of phosphate had no issue on yield and, if in that location's a rapid conversion from polyphosphates to orthophosphates, these results are to be expected. Similar results from other studies have been reported throughout the Corn Belt.
Soil pH should not be an important gene when selecting fertilizer P sources.
From an bookish perspective, monoammonium phosphates (MAP) create a more acidic zone around each fertilizer granule, whereas diammonium phosphates (DAP) create a basic zone. Thus, in high pH soils, we can conjecture that using MAP-based fertilizers should exist better than DAP considering the acrid-producing fertilizer would start the calcareous soils.
An additional concern regarding MAP or DAP pick, bated from soil pH, is potential ammonia toxicity to germinating seeds in dry out soils. In applying the recommended amount of P in a drill-row or popular-up fertilizer placement, DAP will contain approximately lx percent more Due north, which may be a potential injury risk.
However, because agronomic studies and economic data indicate no ingather yield differences, we can conclude that fertilizer option should exist made on traditional factors such as nutrient content, price, availability, etc.
Daniel E. Kaiser, Extension nutrient management specialist and Paulo Pagliari, Extension soil scientist
Acknowledgments
Partial funding for this content was provided past the Metropolitan Council and the Minnesota Lath of Water and Soil Resource.
Reviewed in 2018
Source: https://extension.umn.edu/phosphorus-and-potassium/understanding-phosphorus-fertilizers
Posted by: dorseyaune1973.blogspot.com



0 Response to "What Is Chemical Makeup Of 40 Rock"
Post a Comment